PFAS and Our Educational Portfolio
During my last two months at the Mon Water Project, I have enjoyed a whirlwind of experiences including professional workshops, community outreach, curriculum development, and more! I wanted to update you all on my work so far and ask for any feedback, suggestions, or comments.
This article will also be my first installment in a series of current event articles, posted along with our Newsletter. I hope to bring some attention to various water-based issues facing Pittsburgh and the broader environment around us. If there is anything you would like to learn more about, I am happy to use my time for research and communication!
For the Mon Water Project’s educational programming, I am approaching the process of creating our Educational Portfolio through a lens learned during my time in Dr. Carrie McDonough’s historical micro-pollutant research lab. Our Educational Portfolio has been designed to include this perspective in our tabling activities, one off lessons, full curriculum, field trips, and excursions. We tie existing water science curriculum with urban problems faced locally with goal of bringing the scientific concepts home. We hope that this tool allows interested people to browse what we can offer.
The PFAS Problem
Over two years, I studied as an analytical chemist by identifying and quantifying the concentration of per- and polyfluoroalkyl substances (PFAS) in fish samples from the Hudson River. As I learned more about PFAS, a dynamic connection between water, history, and culture became increasingly elucidated by their presence in our waters today.
The PFAS problem is not only technical, but innately regulatory and cultural. Its history helps has been driven by the chemical industries economic culture and the lack of past regulation. Other environmental issues in our watershed follow similar patterns. Learning about the history and technical aspects of the issue helps to illustrate the underlying culture that created the problem in the first place.
When I approached the technical PFAS research while learning about the more social facets of the public health issue in my free time, I landed flat on an idea that has been present in environmental circles for generations. These problems do not happen in a vacuum. Chemical pollution, and any pollution for that matter is an effect of regulation, culture, and economics. All of this is shaped by our history.
The Chemistry
PFAS are also known as “forever chemicals”, and were accidentally invented in the 1940s. Since manufacturing began, PFAS have been used in a wide variety of products including pots and pans, food wrappers, clothing, furniture, carpets, personal care products, and cleaning products. The PFAS class has grown to include around 14,000 chemicals and counting.
Lately, the news has been focusing more on the public health outcries surrounding PFAS use and exposure. To understand your exposure and the warnings around these chemicals, it is helpful to have an idea of the base chemical structures. For PFAS, I find the chemical structures aid in my understanding of the overall problem.
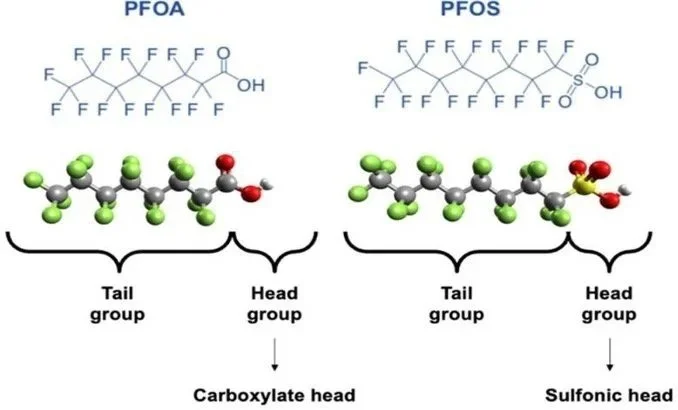
The PFAS class is defined by a specific tail group and varying head group. An example of two of the most common PFAS are shown below in Figure 1. The tail group is a chain of carbon-fluorine bonds, which are some of the shortest and strongest bonds known to chemists. The tail group can change in overall length, with varying amounts of carbons and fluorine. This tail is bonded to a head group, modified based on various desired properties which change based on applications (ie. coatings or flame retardant).
Figure 1: Perfluorooctanoic acid (right) and perfluorooctane sulfonate (left). These are the most popular PFAS produced. Both structures include a carbon-fluorine tail group and varying head group.
The incredibly strong carbon-fluorine bonds give the molecules very high stability and chemical resistance (ie. the bonds are super hard to break in a reaction). Manufacturers love PFAS for these strong bonds. They can keep products clean as they move through factories, make something more thermally stable (like a pan), and are so resistant to degradation that they last “forever” after application.
The reason the chemicals are popular and profitable is also the reason they have become such ubiquitous pollutants. The same tail group that imparts water, oil, chemical, and stain resistance also makes degradation and breakdown in the environment essentially impossible. The inability to degrade and lack of biological, chemical, or thermal breakdown pathways gives the class the name “forever chemicals”. The chemical properties of PFAS also mean the chemicals can move with water, air, and accumulate in soils, fish, and wildlife (learn more). We know that ultimately, PFAS ends up in our bodies, major waterways, and most remote areas on Earth (check this out: The Body, Arctic, and Amazon).
A major concern with exposure to the class of PFAS is that they are toxic to humans in many ways, including liver and cholesterol issues, increased cancer risks, and lower birth weights. Toxic effects from PFAS come from both acute (large doses occurring once) and chronic low dose exposure (small doses occurring over time). In everyday life, chronic low dose toxic exposure to PFAS means exposure to a large class of many chemicals, all at different doses and with different routes of exposure. This combination of variables makes the PFAS issue difficult to tackle on a scientific level. To try and tamp down exposure, scientists have been warning public health officials for decades about PFAS based products, and the combination of PFAS in surface and drinking water.
The Chemical Culture
A terribly not fun fact is that the chemical industry knew PFAS were toxic since the early 1960s and 1970s, about 40 years before public health communities. People working in manufacturing facilities and the communities surrounding these sites were the first hosts to a barrage of health issues and water contamination. The chemical facilities around these communities have been held legally responsible for knowingly harming the local people while manufacturing PFAS products. Some regulators and academics believe that this form of harming for profit is not a specifically PFAS issue, but one derived from a culture in the chemical industry to manufacture and sell first and determine safety later.
The Economics
This backwards culture of thought currently allows the chemical industry to manufacture, sell, and use profits to push down possible safety concerns for economic benefit (also seen with cigarette manufacturers). It is important to note that the chemical industry makes up about 25% of the US GDP. Policy makers also receive funding from the chemical industry in droves. This combination of economic strength and lobbying make regulating PFAS incredibly difficult; chemical manufacturers are still allowed to create and sell PFAS.
But all is not lost! Stakeholders around the world have begun working together to create regulatory solutions that combine public health, scientific knowledge, and economic needs. Efforts have been successful. Take a look at an example in Delaware: 2026 Strategic Framework for Contaminants of Emerging Concern and a 2026 PFAS Implementation Plan. Due to the increase in attention towards PFAS exposure and safety concerns, even the chemical industry is starting to work towards making remediation profitable. So much so that the clean-up of PFAS from the environment is becoming an investment opportunity. Interdisciplinary approaches have led to the creation of applicable and actionable solutions.
The Pittsburgh Part
The PFAS problem is also directly present in Pittsburgh. Three Rivers Waterkeeper, a local environmental organization focused on legal advocacy for the Allegheny, Ohio, and Monongahela have worked hard to sample PFAS in the tributaries around the Pittsburgh International Airport. Their monitoring data has shown high levels of PFAS (100 times the EPA guideline) in Montour Run which flows into the Ohio. This spike in PFAS detection has been attributed to the use of AFFF, a fire fighting foam made up of PFAS. The airport has stated that they no longer use AFFF after the Federal Aviation Administration removed its requirement of use, but PFAS is still being detected (learn more).
The persistence of PFAS in Pittsburgh means that we cannot simply regulate and forget. To move forward proper remediation tactics and continued investigation into PFAS needs to be supported. Part of this fight includes informing our communities of the issue and teaching the science behind the public health warnings.
The Educational Portfolio
The PFAS problem is a truly interdisciplinary issue. In Mon Water Project’s educational portfolio, I hope to use this interdisciplinary approach and our hyper local framework to illustrate the dynamic relationship of water issues and history in Pittsburgh. I have been and will be exploring/teaching the intersection between topics like urban flooding and the history of industrialization, stormwater pollution and the history of railroad construction, as well as macroinvertebrate populations and overall river health. Each of these broad topics provide a connection between Pittsburgh’s unique landscape, history, community, and cultures. My hope is that participants will leave our programming feeling that they are not only more informed about the science, but more informed of the entire scope of an issue.
If you have any questions or comments, please reach out: morgan@monwaterproject.org